Quality of Black-Boned Chicken (Gallus Domesticus) Carcass and Development to Black-Boned Chicken Soup
Main Article Content
บทคัดย่อ
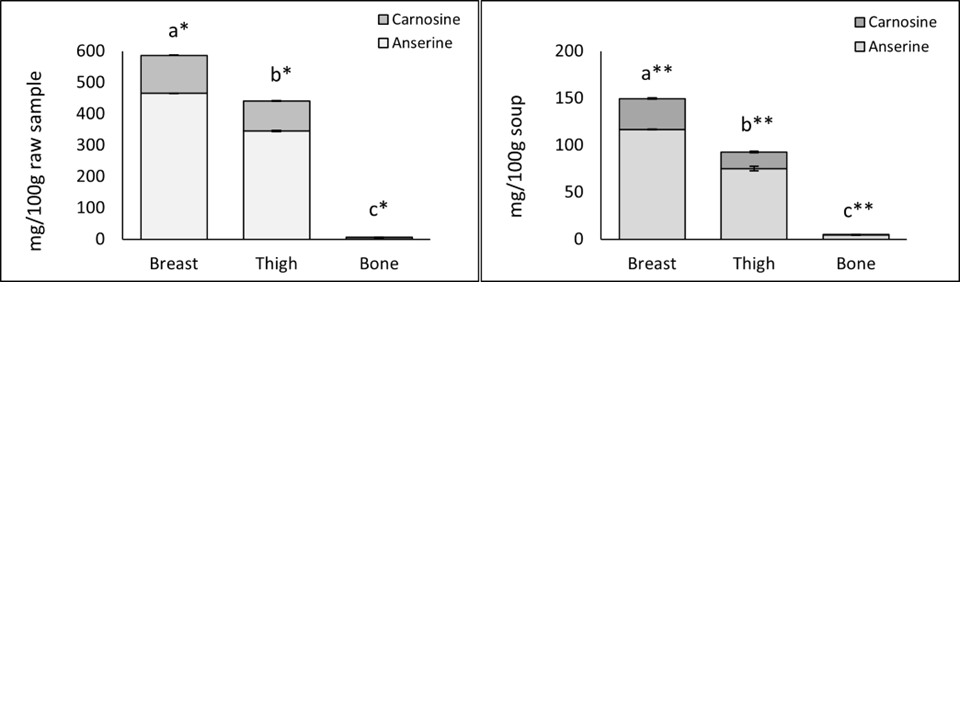
Black-boned chickens are one of the native fowl with distinctive characteristics. It was found that it has high protein and low-fat content. Additionally, it had melanin which is a natural antioxidant compound. Chicken soup is a popular functional food that contains carnosine and anserine. These substances are the most prevalent histidine-containing dipeptides not found in plants. Thus, this study aimed to examine the quality of black-boned chicken carcasses and evaluate the biological properties of melanin, carnosine, and anserine were evaluated in various part of black-boned chicken: breast meat, thigh meat, and femur bone as well as the soups derived from each part. The chicken breast meat and its soup contained more carnosine and anserine than the those found thigh meat and femur bone soups (P<0.05). Although, the melanin content showed different amount in raw samples. The chicken soups were not significantly different (P>0.05). The anserine content in chicken muscles were found to be higher compared to that of carnosine. The biological properties of carnosine and anserine extracts from chicken muscle increased proportionally with the amount of these peptides in the muscle. Meanwhile, the femur bone had fewer peptides than the muscle. The femur bone exhibited higher antioxidant activities than the chicken thigh soup, as measured by ABTS and FRAP assays. Hence, the antioxidant activities corresponding with the dipeptides except femur bone.
Article Details

อนุญาตภายใต้เงื่อนไข Creative Commons Attribution-NoDerivatives 4.0 International License.
บทความที่ได้รับการตีพิมพ์เป็นลิขสิทธิ์ของวารสาร มรภ.กพ. วิทยาศาสตร์ คณิตศาสตร์ และเทคโนโลยี
ข้อคิดเห็นใดๆ ที่ปรากฎในวารสารเป็นวรรณกรรมของผู้เขียนโดยเฉพาะ ซึ่งมหาวิทยาลัยราชภัฏกำแพงเพชรและบรรณาธิการไม่จำเป็นต้องเห็นด้วย
เอกสารอ้างอิง
Ponnarong Prasertsri. (2022). Thailand: Poultry and Products Annual.
Jaturasitha, S., Srikanchai, T., Kreuzer, M., & Wicke, M. (2008). Differences in carcass and meat characteristics between chicken indigenous to northern Thailand (Black-boned and Thai native) and imported extensive breeds (Bresse and Rhode Island red). Poult Sci, 87(1), 160-169. https://doi.org/10.3382/ps.2006-00398
Muroya, S., Tanabe, R.-I., Nakajima, I., & Chikuni, K. (2000). Molecular Characteristics and Site Specific Distribution of the Pigment of the Silky Fowl. The Journal of veterinary medical science / the Japanese Society of Veterinary Science, 62, 391-395. https://doi.org/10.1292/jvms.62.391
Hsieh, P.-H., & Lien, T.-F. (2012). Study of the Physico-chemical Properties and Antioxidant Activity of Extracted Melanins https://www.ccsenet.org/journal/index.php/jas/article/view/17855
Tian, Y., Xie, M., Wang, W., Wu, H., Fu, Z., & Lin, L. (2007). Determination of carnosine in Black-Bone Silky Fowl (Gallus gallus domesticus Brisson) and common chicken by HPLC. European Food Research and Technology, 226, 311-314. https://doi.org/10.1007/s00217-006-0528-1
Jung, S., Bae, Y. S., Kim, H. J., Jayasena, D. D., Lee, J. H., Park, H. B., Heo, K. N., & Jo, C. (2013). Carnosine, anserine, creatine, and inosine 5′-monophosphate contents in breast and thigh meats from 5 lines of Korean native chicken. Poultry Science, 92(12), 3275-3282. https://doi.org/10.3382/ps.2013-03441
Chen, S.-R., Jiang, B., Zheng, J.-X., Xu, G.-Y., Li, J.-Y., & Yang, N. (2008). Isolation and characterization of natural melanin derived from silky fowl (Gallus gallus domesticus Brisson). Food Chemistry, 111(3), 745-749. https://doi.org/10.1016/j.foodchem.2008.04.053
Tadesse, S. A., & Emire, S. A. (2020). Production and processing of antioxidant bioactive peptides: A driving force for the functional food market. Heliyon, 6(8), e04765-e04765. https://doi.org/10.1016/j.heliyon.2020.e04765
Monica, B., & Ioan, S. (2019). Functional Food. International Journal of Nutrition, 3(3), 7-16. https://doi.org/10.14302/issn.2379-7835.ijn-19-2615
Martini, S., Solieri, L., & Tagliazucchi, D. (2021). Peptidomics: new trends in food science. In (Vol. 39, pp. 51-59): Elsevier Ltd.
Jaturasitha, S., Chaiwang, N., & Kreuzer, M. (2016). Thai native chicken meat: an option to meet the demands for specific meat quality by certain groups of consumers; a review. Animal Production Science, 57. https://doi.org/10.1071/AN15646
Chan, L., Wang, H.-M., Chen, K.-Y., Lin, Y.-C., Wu, P.-J., Hsieh, W.-L., Chen, Y.-R., Liu, C.-P., Tsai, H.-Y., Chen, Y.-R., Chang, H.-H., Hsieh, Y.-C., & Hu, C.-J. (2016). Effectiveness of Essence of Chicken in Improving Cognitive Function in Young People Under Work-Related Stress: A Randomized Double-Blind Trial. Medicine, 95(19). https://doi.org/10.1097/MD.0000000000003640
Li, Y. F., He, R. R., Tsoi, B., & Kurihara, H. (2012). Bioactivities of Chicken Essence. Journal of Food Science, 77(4), R105-R110. https://doi.org/10.1111/j.1750-3841.2012.02625.x
Yamano, E., Tanaka, M., Ishii, A., Tsuruoka, N., Abe, K., & Watanabe, Y. (2013). Effects of chicken essence on recovery from mental fatigue in healthy males https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3707410/
Lü, Y. Q., He, R. R., Watanabe, H., Abe, K., Sakurai, E., Yanai, K., & Kurihara, H. (2010). Effects of a chicken extract on food-deprived activity stress in rats. Biosci Biotechnol Biochem, 74(6), 1276-1278. https://doi.org/10.1271/bbb.90950
Konagai, C., Watanabe, H., Abe, K., Tsuruoka, N., & Koga, Y. (2013). Effects of Essence of Chicken on Cognitive Brain Function: A Near-Infrared Spectroscopy Study. Bioscience, biotechnology, and biochemistry, 77. https://doi.org/10.1271/bbb.120706
Dong, Z. Y., Li, M. Y., Tian, G., Zhang, T. H., Ren, H., & Quek, S. Y. (2019). Effects of ultrasonic pretreatment on the structure and functionality of chicken bone protein prepared by enzymatic method. Food Chemistry, 299, 125103-125103. https://doi.org/10.1016/j.foodchem.2019.125103
AOAC. (2000). Official methods of analysis of AOAC International (17th ed.). AOAC International.
Mora, L., Sentandreu, M. A., & Toldrá, F. (2007). Hydrophilic chromatographic determination of carnosine, anserine, balenine, creatine, and creatinine. J Agric Food Chem, 55(12), 4664-4669. https://doi.org/10.1021/jf0703809
Lin, L.-C., & Chen, W.-T. (2005). The Study of Antioxidant Effects in Melanins Extracted from Various Tissues of Animals. Asian-Australasian Journal of Animal Sciences, 18. https://doi.org/10.5713/ajas.2005.277
Tanzadehpanah, H., Asoodeh, A., & Chamani, J. (2012). An antioxidant peptide derived from Ostrich (Struthio camelus) egg white protein hydrolysates. Food Research International, 49, 105–111. https://doi.org/10.1016/j.foodres.2012.08.022
Benzie, I. F., & Strain, J. J. (1996). The ferric reducing ability of plasma (FRAP) as a measure of "antioxidant power": the FRAP assay. Anal Biochem, 239(1), 70-76. https://doi.org/10.1006/abio.1996.0292
Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M., & Rice-Evans, C. (1999). Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med, 26(9-10), 1231-1237. https://doi.org/10.1016/s0891-5849(98)00315-3
Cansu, Ü., & Boran, G. (2015). Optimization of a Multi-Step Procedure for Isolation of Chicken Bone Collagen. Korean J Food Sci Anim Resour, 35(4), 431-440. https://doi.org/10.5851/kosfa.2015.35.4.431
Barido, F. H., Lee, C. W., Park, Y. S., Kim, D. Y., & Lee, S. K. (2021). The effect of a finishing diet supplemented with γ-aminobutyric acids on carcass characteristics and meat quality of Hanwoo steers. Anim Biosci, 34(4), 621-632. https://doi.org/10.5713/ajas.20.0224
Lawrie, R. A., & Ledward, D. (2014). Lawrie’s meat science. Woodhead Publishing.
Listrat, A., Lebret, B., Louveau, I., Astruc, T., Bonnet, M., Lefaucheur, L., Picard, B., & Bugeon, J. (2016). How Muscle Structure and Composition Influence Meat and Flesh Quality. ScientificWorldJournal, 2016, 3182746. https://doi.org/10.1155/2016/3182746
Ionescu, R. E., Fillit, C., Jaffrezic-Renault, N., & Cosnier, S. (2008). Urease–gelatin interdigitated microelectrodes for the conductometric determination of protease activity. Biosensors and Bioelectronics, 24(3), 489-492. https://doi.org/10.1016/j.bios.2008.06.021
Bee, S.-L., & Hamid, Z. A. A. (2019). Characterization of chicken bone waste-derived hydroxyapatite and its functionality on chitosan membrane for guided bone regeneration. Composites Part B: Engineering, 163, 562-573. https://doi.org/10.1016/j.compositesb.2019.01.036
Lesiów, T. (2000). Gelation of chicken breast and thigh muscle homogenates: Effect of pH and time of aging. Food / Nahrung, 44(6), 426-430. https://doi.org/10.1002/1521-3803(20001201)44:6<426::AID-FOOD426>3.0.CO;2-Y
Soeparno. (2009). Ilmu dan Teknologi Daging (2 ed.).
Jaturasitha, S. (2012). Meat Technology. Mingmuang Press.
Barido, F. H., Kang, S. M., & Lee, S. K. (2022). The Quality and Functional Improvement of Retorted Korean Ginseng Chicken Soup (Samgyetang) by Enzymolysis Pre-Treatment with Cordyceps militaris Mushroom Extract. Foods, 11(3), 422. https://doi.org/10.3390/foods11030422
Paião, F. G., Ferracin, L. M., Pedrão, M., Kato, T., & Shimokomaki, M. (2013). Skeletal muscle calcium channel ryanodine and the development of pale, soft, and exudative meat in poultry. Genet Mol Res, 12(3), 3017-3027. https://doi.org/10.4238/2013.August.20.3
Ang, S. S., & Ismail-Fitry, M. R. (2019). Production of Different Mushroom Protein Hydrolysates as Potential Flavourings in Chicken Soup Using Stem Bromelain Hydrolysis. Food Technol Biotechnol, 57(4), 472-480. https://doi.org/10.17113/ftb.57.04.19.6294
Li, Q., Liu, J., De Gobba, C., Zhang, L., Bredie, W. L. P., & Lametsch, R. (2020). Production of Taste Enhancers from Protein Hydrolysates of Porcine Hemoglobin and Meat Using Bacillus amyloliquefaciens γ-Glutamyltranspeptidase. Journal of Agricultural and Food Chemistry, 68(42), 11782-11789. https://doi.org/10.1021/acs.jafc.0c04513
Kojima, S., Saegusa, H., & Sakata, M. (2014). Histidine-Containing Dipeptide Concentration and Antioxidant Effects of Meat Extracts from Silky Fowl: Comparison with Meat-Type Chicken Breast and Thigh Meats. Food Science and Technology Research, 20(3), 621-628. https://doi.org/10.3136/fstr.20.621
Peiretti, P. G., Medana, C., Visentin, S., Giancotti, V., Zunino, V., & Meineri, G. (2011). Determination of carnosine, anserine, homocarnosine, pentosidine and thiobarbituric acid reactive substances contents in meat from different animal species. Food Chem, 126(4), 1939-1947. https://doi.org/10.1016/j.foodchem.2010.12.036
Peiretti, P. G., Medana, C., Visentin, S., Dal Bello, F., & Meineri, G. (2012). Effect of cooking method on carnosine and its homologues, pentosidine and thiobarbituric acid-reactive substance contents in beef and turkey meat. Food Chemistry, 132(1), 80-85. https://doi.org/10.1016/j.foodchem.2011.10.035
Ustunol, Z. (2014). Physical, Chemical, and Processing-Induced Changes in Proteins. In Applied Food Protein Chemistry (pp. 23-46). https://doi.org/10.1002/9781118860588.ch4
Chan, K. M., & Decker, E. A. (1994). Endogenous skeletal muscle antioxidants. Crit Rev Food Sci Nutr, 34(4), 403-426. https://doi.org/10.1080/10408399409527669
Stegen, S., Everaert, I., Deldicque, L., Vallova, S., de Courten, B., Ukropcova, B., Ukropec, J., & Derave, W. (2015). Muscle histidine-containing dipeptides are elevated by glucose intolerance in both rodents and men. PLoS One, 10(3), e0121062. https://doi.org/10.1371/journal.pone.0121062
Xin, C., Ma, J. H., Tan, C. J., Yang, Z., Ye, F., Long, C., Ye, S., & Hou, D. B. (2015). Preparation of melanin from Catharsius molossus L. and preliminary study on its chemical structure. J Biosci Bioeng, 119(4), 446-454. https://doi.org/10.1016/j.jbiosc.2014.09.009
Sawhney, S. S. (1994). Thermal stability of melanin. Thermochimica Acta, 247(2), 377-380.
Serpen, A., Gökmen, V., & Fogliano, V. (2012). Total antioxidant capacities of raw and cooked meats. Meat Sci, 90(1), 60-65. https://doi.org/10.1016/j.meatsci.2011.05.027
Chen, H.-M., Muramoto, K., Yamauchi, F., & Nokihara, K. (1996). Antioxidant Activity of Designed Peptides Based on the Antioxidative Peptide Isolated from Digests of a Soybean Protein. Journal of Agricultural and Food Chemistry, 44(9), 2619-2623. https://doi.org/10.1021/jf950833m
Chen, H. M., Muramoto, K., Yamauchi, F., Fujimoto, K., & Nokihara, K. (1998). Antioxidative Properties of Histidine-Containing Peptides Designed from Peptide Fragments Found in the Digests of a Soybean Protein. J Agric Food Chem, 46(1), 49-53. https://doi.org/10.1021/jf970649w
Theodore, A. E., Raghavan, S., & Kristinsson, H. G. (2008). Antioxidative activity of protein hydrolysates prepared from alkaline-aided channel catfish protein isolates. J Agric Food Chem, 56(16), 7459-7466. https://doi.org/10.1021/jf800185f
Gu, F.-L., Kim, J. M., Abbas, S., Zhang, X.-M., Xia, S.-Q., & Chen, Z.-X. (2010). Structure and antioxidant activity of high molecular weight Maillard reaction products from casein–glucose. Food Chemistry, 120(2), 505-511. https://doi.org/10.1016/j.foodchem.2009.10.044
Hwang, I. G., Kim, H. Y., Woo, K. S., Lee, J., & Jeong, H. S. (2011). Biological activities of Maillard reaction products (MRPs) in a sugar–amino acid model system. Food Chemistry, 126(1), 221-227. https://doi.org/10.1016/j.foodchem.2010.10.103