Antibacterial Activities of Palmitic and Lauric Acids from Palm Kernel Oil for the Development of Food-Grade Disinfectants
Main Article Content
บทคัดย่อ
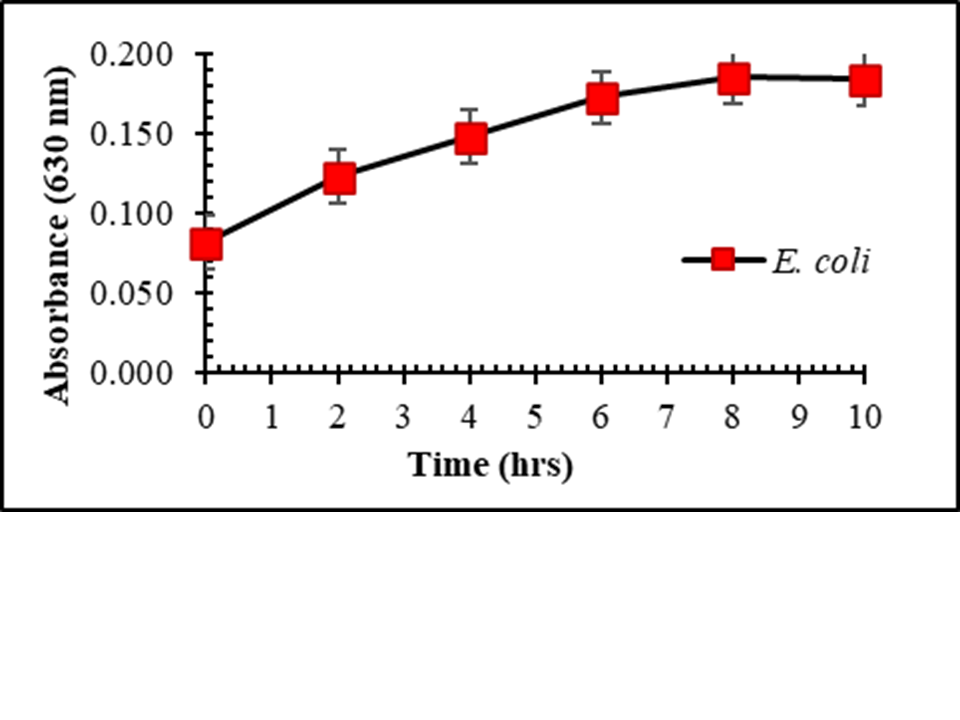
Palmitic and lauric acids, derived from palm kernel oil, exhibit antibacterial activities that are less harmful to humans. This study aimed to evaluate the antibacterial activities and optimal concentrations of palmitic and lauric acids against Staphylococcus aureus and Escherichia coli using the agar well diffusion method, with the goal of developing a food-grade disinfectant. The results demonstrated that a ratio of 87.5:175 mg/ml of palmitic acid to lauric acid was the optimal mix for inhibiting the growth of Staphylococcus aureus and Escherichia coli. The average size of the inhibition zone was 10.40±0.57, 10.59±1.08, 10.57±0.40, 8.67±0.98, 6.38±0.29, and 10.98±0.59 mm. The MIC ratios (200, 62.5, 87.5 mg/ml) and MBC ratios (50, 62.5, 87.5 mg/ml) of palmitic acid combined with lauric acid were 800, 125, 175 mg/ml and 200, 250, 175 mg/ml, respectively. The study found that the combination of palmitic and lauric acids effectively inhibited S. aureus and E. coli. Furthermore, these acids could be used to disinfect food and reduce the reliance on chemical disinfectants in food manufacturing cleaning processes.
Article Details

อนุญาตภายใต้เงื่อนไข Creative Commons Attribution-NoDerivatives 4.0 International License.
บทความที่ได้รับการตีพิมพ์เป็นลิขสิทธิ์ของวารสาร มรภ.กพ. วิทยาศาสตร์ คณิตศาสตร์ และเทคโนโลยี
ข้อคิดเห็นใดๆ ที่ปรากฎในวารสารเป็นวรรณกรรมของผู้เขียนโดยเฉพาะ ซึ่งมหาวิทยาลัยราชภัฏกำแพงเพชรและบรรณาธิการไม่จำเป็นต้องเห็นด้วย
เอกสารอ้างอิง
Langsrud, S., Sidhu, M. S., Heir, E., & Holck, A. L. (2003). Bacterial disinfectant resistance—a challenge for the food industry. International Biodeterioration & Biodegradation, 51(4), 283-290. https://doi.org/https://doi.org/10.1016/S0964-8305(03)00039-8
Akpomie, O. O., Akponah, E., Ehwarieme, A. D., & Paul, R. E. (2020). Antimicrobial activity of coconut water, oil and palm kernel oils extracted from coconut and palm kernel on some plasmid-mediated multi-drug resistant organisms associated with food spoilage. African Journal of Microbiology Research, 14(7), 366-373. doi: 10.5897/AJMR2020.9363
Colchester, M., Forest Peoples Programme & Sawit Watch. (2011) Oil palm expansion in South East Asia trends and implications for local communities and indigenous peoples. Bogor, West Java, Indonesia: Perkumpulan Sawit Watch; England: Forest Peoples Programme. [Pdf] Retrieved from the Library of Congress, https://www.loc.gov/item/2012330582/
Febrina, D., Febriyanti, R., Ikhsan Zam, S., Handoko, J., Fatah, A. E., & Juliantoni, J. (2018). Antibacterial Activity Testing and Ethanol Extract Characterization of Oil Palm Fronds (Elaeis guineensis Jacq). Pakistan Journal of Nutrition.
Hovorková, P., Laloučková, K., & Skřivanová, E. (2018). Determination of in vitro antibacterial activity of plant oils containing medium-chain fatty acids against Gram-positive pathogenic and gut commensal bacteria. Czech Journal of Animal Science, 63(No. 3), 119-125. https://doi.org/10.17221/70/2017-cjas
Huang, C. B., Alimova, Y., Myers, T. M., & Ebersole, J. L. (2011). Short- and medium-chain fatty acids exhibit antimicrobial activity for oral microorganisms. Arch Oral Biol, 56(7), 650-654. https://doi.org/10.1016/j.archoralbio.2011.01.011
Nitbani, F. O., Jumina, Siswanta, D., & Solikhah, E. N. (2016). Isolation and Antibacterial Activity Test of Lauric Acid from Crude Coconut Oil (Cocos nucifera L.). Procedia Chemistry, 18, 132-140. https://doi.org/10.1016/j.proche.2016.01.021
Nguyen, L. N., Trofa, D., & Nosanchuk, J. D. (2009). Fatty acid synthase impacts the pathobiology of Candida parapsilosis in vitro and during mammalian infection. PLoS One, 4(12), e8421. https://doi.org/10.1371/journal.pone.0008421
Nguyen, L. N., & Nosanchuk, J. D. (2011). Lipid droplet formation protects against gluco/lipotoxicity in Candida parapsilosis: an essential role of fatty acid desaturase Ole1. Cell Cycle, 10(18), 3159-3167. https://doi.org/10.4161/cc.10.18.16932
Ray, P. D., Huang, B. W., & Tsuji, Y. (2012). Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal, 24(5), 981-990. https://doi.org/10.1016/j.cellsig.2012.01.008
Desbois, A. P., & Smith, V. J. (2010). Antibacterial free fatty acids: activities, mechanisms of action and biotechnological potential. Appl Microbiol Biotechnol, 85(6), 1629-1642. https://doi.org/10.1007/s00253-009-2355-3
Karimi, E., Jaafar, H. Z., Ghasemzadeh, A., & Ebrahimi, M. (2015). Fatty acid composition, antioxidant and antibacterial properties of the microwave aqueous extract of three varieties of Labisia pumila Benth. Biol Res, 48(1), 9. https://doi.org/10.1186/0717-6287-48-9
Auttajinda N., R., P., Klangpetch U. W., Jirarattanarangsri W. & Osiriphun S. (2022). Palmitic Acid Concentration Acts as an Antibacterial Activity. Food and Applied Bioscience International Conference (FAB 2022), 96. https://www.agro.cmu.ac.th/FAB2022/Home/Home.aspx?MenuID=3
McGaw, L. J., Jäger, A. K., & van Staden, J. (2002). Isolation of antibacterial fatty acids from Schotia brachypetala. Fitoterapia, 73(5), 431–433. https://doi.org/10.1016/s0367-326x(02)00120-x
Goy, R. C., Morais, S. T. B., & Assis, O. B. G. (2016). Evaluation of the antimicrobial activity of chitosan and its quaternized derivative on E. coli and S. aureus growth. Revista Brasileira de Farmacognosia, 26(1), 122-127. https://doi.org/10.1016/j.bjp.2015.09.010
CLSI. (2020). Performance Standards for Antimicrobial Susceptibility Testing. 30th ed. CLSI supplement M100 Wayne, PA: Clinical and Laboratory Standards Institute.
Rodriguez-Melcon, C., Alonso-Calleja, C., Garcia-Fernandez, C., Carballo, J., & Capita, R. (2021). Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC) for Twelve Antimicrobials (Biocides and Antibiotics) in Eight Strains of Listeria monocytogenes. Biology (Basel), 11(1). https://doi.org/10.3390/biology11010046
Wang, L., Fan, D., Chen, W., & Terentjev, E. M. (2015). Bacterial growth, detachment and cell size control on polyethylene terephthalate surfaces. Sci Rep, 5, 15159. https://doi.org/10.1038/srep15159
Fujikawa, H., & Morozumi, S. (2005). Modeling surface growth of Escherichia coli on agar plates. Appl Environ Microbiol, 71(12), 7920-7926. https://doi.org/10.1128/aem.71.12.7920-7926.2005
Maier, R. M., & Pepper, I. L. (2015). Chapter 3 - Bacterial Growth. In I. L. Pepper, C. P. Gerba, & T. J. Gentry (Eds.), Environmental Microbiology (Third Edition) (pp. 37-56). Academic Press. https://doi.org/https://doi.org/10.1016/B978-0-12-394626-3.00003-X
Bantawa, K., Sah, S. N., Subba Limbu, D., Subba, P., & Ghimire, A. (2019). Antibiotic resistance patterns of Staphylococcus aureus, Escherichia coli, Salmonella, Shigella and Vibrio isolated from chicken, pork, buffalo and goat meat in eastern Nepal. BMC research notes, 12(1), 766. https://doi.org/10.1186/s13104-019-4798-7
Acsa, I., Lilly Caroline, B., Philip Njeru, N., & Lucy Wanjiru, N. (2021). Preliminary Study on Disinfectant Susceptibility/Resistance Profiles of Bacteria Isolated from Slaughtered Village Free-Range Chickens in Nairobi, Kenya. International journal of microbiology, 2021, 8877675. https://doi.org/10.1155/2021/8877675
McGaw, L. J., Jäger, A. K., & van Staden, J. (2002). Antibacterial effects of fatty acids and related compounds from plants. South African Journal of Botany, 68(4), 417-423. https://doi.org/10.1016/s0254-6299(15)30367-7
Seidel, V., & Taylor, P. W. (2004). In vitro activity of extracts and constituents of against rapidly growing mycobacteria. International journal of antimicrobial agents, 23(6), 613–619. https://doi.org/10.1016/j.ijantimicag.2003.11.008
Agoramoorthy, G., Chandrasekaran, M., Venkatesalu, V., & Hsu, M.J. (2007). Antibacterial and antifungal activities of fatty acid methyl esters of the blind-your-eye mangrove from India. Brazilian Journal of Microbiology, 38, 739-742. https://doi.org/ 10.1590/S1517-83822007000400028
Ekpa, O. D. E., R.U.B. (1996). Comparative studies of mmanyanga, palm and coconut oils: antimicrobial effects of the oils and their metallic soaps on some bacteria and fungi. Global Journal of Pure and Applied Sciences, 2(2), 155-163. https://eurekamag.com/research/002/782/002782965.php
Ekwenye, U. N. I., C.A. (2005). Antimicrobial effects of palm kernel oil and palm oil. Current Applied Science and Technology, 5, 502-505. https://li01.tci-thaijo.org/index.php/cast/article/view/166606
Al-Akhras, M. A., Alshorman, M., Masadeh, M., Aljarrah, K., & Ababneh, Z. (2018). Modeling the effect of Rose Bengal on growth and decay patterns of Pseudomonas aeruginosa, Escherichia coli and Staphylococcus aureus. IOP Conference Series: Materials Science and Engineering, 305, 012004. https://doi.org/10.1088/1757-899X/305/1/012004
Sheikh, A. A., Checkley, S., Avery, B., Chalmers, G., Bohaychuk, V., Boerlin, P., Reid-Smith, R., & Aslam, M. (2012). Antimicrobial resistance and resistance genes in Escherichia coli isolated from retail meat purchased in Alberta, Canada. Foodborne pathogens and disease, 9(7), 625–631. https://doi.org/10.1089/fpd.2011.1078
Putri, N. I. C. A., Ramadhani, R., & Wasito, E. B. (2021). Gram Negative Bacteria (Escherichia coli) Win Against Gram Positive Bacteria (Staphylococcus aureus) in The Same Media. Biomolecular and Health Science Journal. https://doi.org/10.20473/bhsj.v4i2.30177